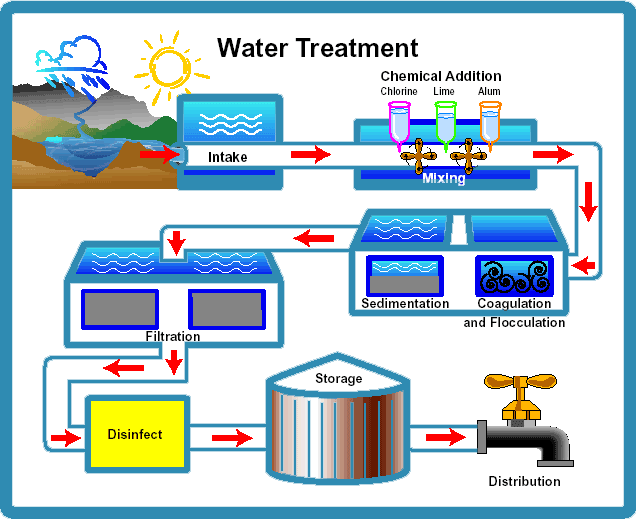
Generally, there are about five steps commonly used in the municipal wastewater treatment process; chemical coagulation, flocculation, sedimentation, filtration, and disinfection.
Disinfection can destroy microorganisms that may cause diseases in humans. It is an essential step in the water treatment process. Public water systems utilize various disinfection practices, some of which can potentially create undesired, harmful chemical byproducts.
The U.S. Environmental Protection Agency (EPA) allows drinking water treatment plants to use chloramine and chlorine to disinfect drinking water. Research shows that chloramine and chlorine both have benefits and drawbacks.
Chlorine is a highly effective method of disinfection and does a great job eliminating most pathogens from the water we drink. However, it produces small amounts of chemicals (called “disinfection by-products”) while in the pipes if the source water has higher levels of dirt or germs, they may react with the chlorine 1. These disinfection by-products can cause illness after drinking them for a long time.
Also, chlorine is used up quickly in water systems. Sometimes there is not enough chlorine left to kill germs in the water by the time it reaches the end of the pipes where it goes from the drinking water distribution system to the end user.
Chloramine is a mixture of ammonia and chlorine. Monochloramine is the most widely used form of chloramine. This compound can last longer in the water pipes and produces fewer disinfection by-products 1. However, studies indicate that chloramine causes more rapid deterioration of the municipal infrastructure. In water systems that still use lead pipes or components this causes lead and other metals to leach into drinking water.
In 1998, an EPA survey estimated 68 million Americans were drinking water disinfected with chloramine. Several major U.S. cities such as Philadelphia, San Francisco, Tampa Bay, and Washington, D.C. use chloramine to disinfect drinking water.2 It does not work as effectively as chlorine.
Nitrification is a side effect of using chloramine. It occurs when small amounts of excess ammonia promote the growth of “nitrifying” bacteria in the distribution system. Nitrification increases when the water temperature rises. Nitrifying bacteria can be difficult to eradicate and cause water quality problems. These bacteria can deplete chlorine in the water, raising the risk of waterborne illness if chlorine levels are not monitored and adjusted.
We will cover more about nitrates, nitrites and nitrification in Part 2 of our discussion about the water treatment process. Aquaspace water filters are currently being used by the Washington, D,C. school system to address concerns about lead in drinking water. Annual testing of the filters by the DC school system showed that our filters continue to be an effective deterrent for lead in the drinking water. For more information about standards for drinking water treatment see the EPA website www.epa.gov.
Footnote 1 EPA. Information about chloramine in drinking water,2012.
Footnote 2 EPA. Basic information about Chloramines and Drinking Water Disinfection Chloramine 2013.



Leave a comment